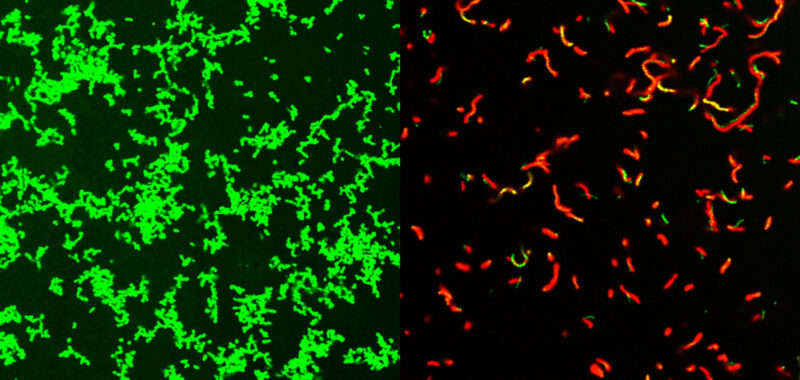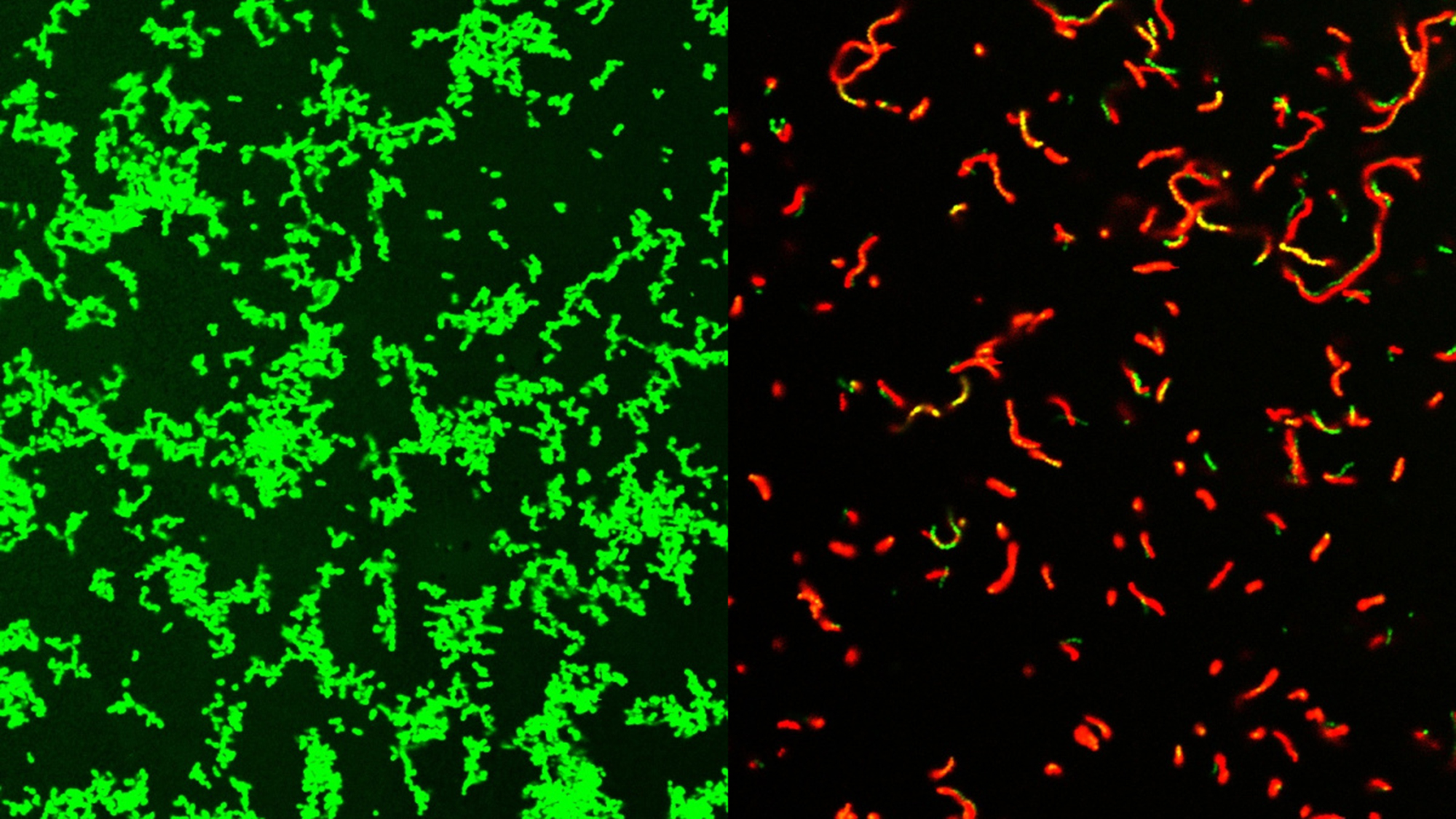
An international team of scientists has developed a new family of compounds that can clear bacterial infections in mice. Some of these infections can result in serious “flesh-eating” illnesses. There are about 700 to 1,100 cases of flesh-eating illnesses every year in the United States. The new family of compounds could also represent the beginning of a new class of antibiotics and are described in a study published August 2 in the journal Science Advances.
Growing resistance
For decades, clinicians have been sounding the alarm about pathogens that are increasingly becoming more resistant to drugs currently available. This makes them more dangerous and according to the Centers for Disease Control and Prevention (CDC), over 2.8 million antimicrobial-resistant infections occur in the US every year. More than 35,000 people die from these infections. To combat this, newer antimicrobial compounds will be needed to replace the ones that bacteria have become resistant to.
Molecular microbiologists Scott Hultgren and Michael Caparon from Washington University School of Medicine in St. Louis and chemist Fredrik Almqvist from the University of Umeå in Sweden collaborated on this new family of compounds called GmPcides.
[Related: These flesh-eating bacteria are finding new beaches to call home.]
GmPcides work by targeting gram-positive bacteria. These types of bacteria can cause various drug-resistant staph infections, toxic shock syndrome, and other bacterial illnesses that can turn deadly.
“All of the gram-positive bacteria that we’ve tested have been susceptible to that compound. That includes enterococci, staphylococci, streptococci, C. difficile, which are the major pathogenic bacteria types,” Caparon said in a statement. “The compounds have broad-spectrum activity against numerous bacteria.”
A ‘happy accident’
The new GmPcide compounds are based on a type of molecule called ring-fused 2-pyridone that was developed by what the team calls a happy accident. Caparon and Hultgren had asked Almqvist to develop a chemical compound that can prevent bacterial films from latching onto the surface of urethral catheters. These are a common cause of urinary tract infections in hospital settings.
The resulting compound also had infection-fighting properties against multiple types of bacteria. Some of their earlier research showed that GmPcides can kill bacteria strains in petri dish experiments.
In this new study, they took those petri dish experiments one step further by testing how compounds work on necrotizing soft-tissue infections. These fast-spreading infections usually involve multiple types of gram-positive bacteria. Necrotizing fasciitis–or flesh-eating disease–is the best known of these infections. It can rapidly damage tissue so severely that limb amputation is often necessary to control its spread. Roughly 20 percent of patients with flesh-eating disease die.
The team focused on one pathogen that is responsible for about 500,000 deaths every year–Streptococcus pyogenes. A group of mice was infected with S. pyogenes. One group was treated with GmPcide, while the other wasn’t. Those that received the GmPcide treatment fared better than the untreated mice in almost every metric. They lost less weight, had smaller ulcers, and fought off the infection faster. Damaged areas of skin also appeared to heal quicker post-infection.
While it is still not fully clear how GmPcides did all of this, a microscopic examination showed that the treatment has a significant effect on bacterial cell membranes. These are the outer wrapping of the microbes.
[Related: ‘Bacterial glitter’ shimmers without pigments.]
“One of the jobs of a membrane is to exclude material from the outside,” Caparon said. “We know that within five to ten minutes of treatment with GmPcide, the membranes start to become permeable and allow things that normally should be excluded to enter into the bacteria, which suggests that those membranes have been damaged.”
This can alter the bacteria’s own functions, including actions that damage the host and make the bacteria less effective at taking down the host’s immune response to infections.
GmPcides also may be less likely to lead to drug-resistant strains. The experiments designed to create resistant bacteria found that very few cells can withstand treatment. This means they are less likely to pass on their advantages to the next generation of bacteria.
The road ahead
According to Caparon, there are still numerous steps before GmPcides will be available at your local pharmacy. The team has patented the compound used and licensed it to QureTech Bio, a company that Caparon, Hultgren and Almqvist have an ownership stake in. The license was contingent on the expectation that they will collaborate with a separate company that can manage the pharmaceutical development and clinical trials to bring it to market.
According to the team, the kind of collaborative science that created GmPcides will be needed to treat the problems like antimicrobial resistance.
“Bacterial infections of every type are an important health problem, and they are increasingly becoming multi-drug resistant and thus harder to treat,” Hultgren said in a statement. “Interdisciplinary science facilitates the integration of different fields of study that can lead to synergistic new ideas that have the potential to help patients.”